Key analytics and exclusive content under the tree. You can even give someone a last-minute gift with a 1-year Portfolio Signature subscription. And if you buy an annual subscription for multiple friends, friends, and family at the same time, you have a choice Discount on quantitiesRe is. So in addition to making a useful holiday gift, you can also support the production of high-quality economical content.
know more
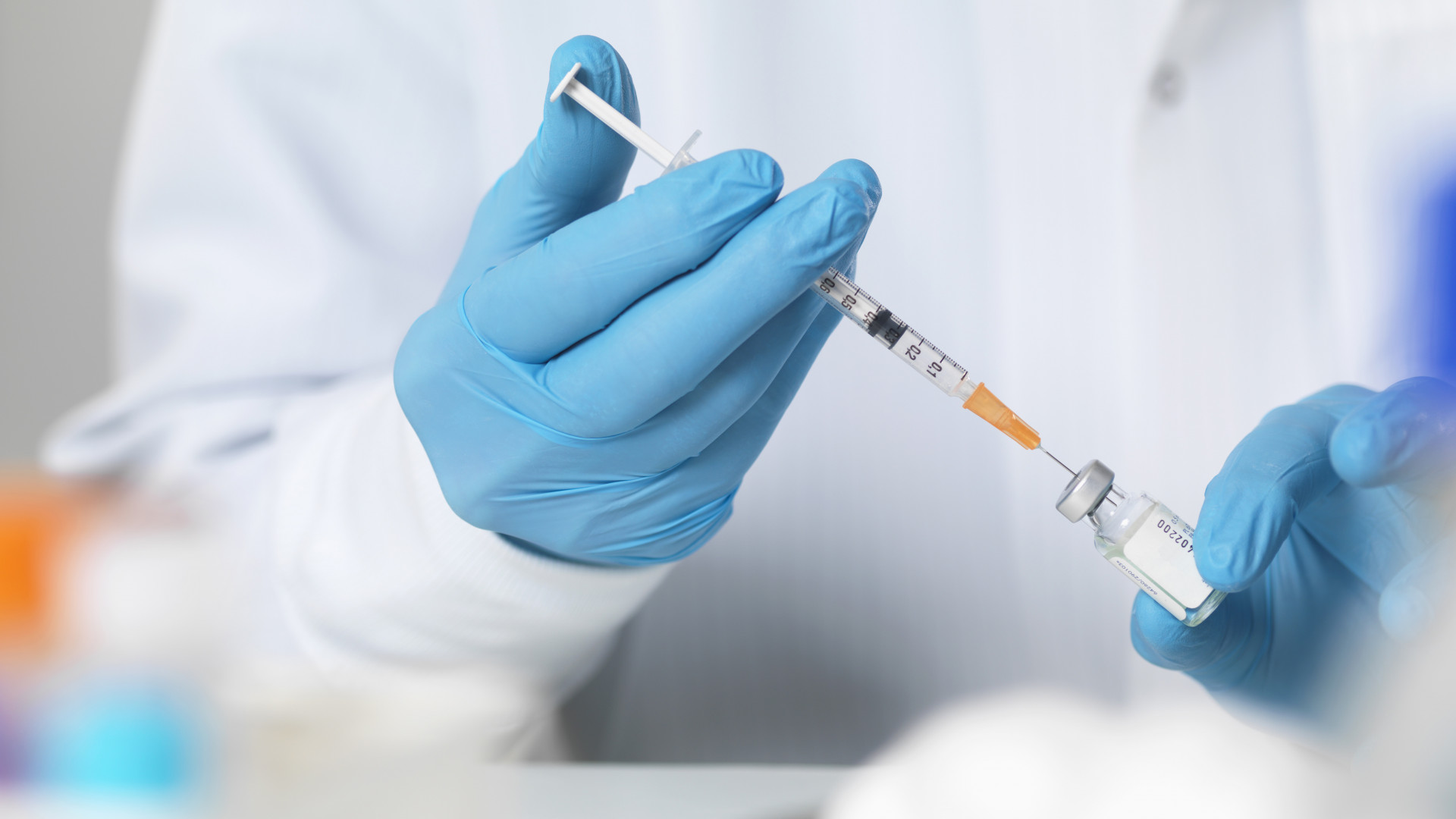
“The safety and efficacy of the vaccine was not studied at different dosing times, and the majority of the participants in the trial received the second dose at specific intervals in the study.”
We have no evidence of continued protection 21 days after the first dose
The joint statement confirmed.
For complete protection, the maximum duration of 42 days for the Pfizer / BioNTech vaccine must be adhered to.
The European News Agency, which authorized the vaccination on December 21, said Monday.
Today, Monday, Germany discussed delaying the administration of the second dose of the vaccine, studying the British model, while Denmark decided to extend the period between giving the two vaccines to six weeks.
Spanish Health Minister Ella Salvador said on Monday that the Spanish Public Health Authority will provide two doses of the BioNTech / Pfizer vaccine within a period to be determined by the manufacturers.
In the United Kingdom it has been decided that the doses that were originally intended for the second dose will also be given as a first dose so that as many people as possible can receive it as soon as possible. The second dose is given 12 weeks later.
Cover photo: Getty Images